
 |
A humidification chamber for relaxing works of art |
Here is a method of humidification that was developed by Tim Padfield and Poul Larsen as part of an experiment in desalting bricks. It is, however, of general usefulness.
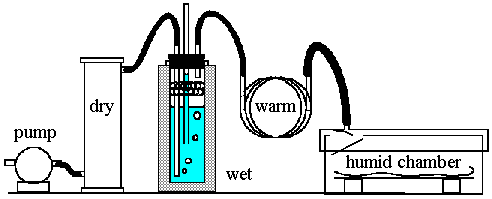
A compressor, air flask or aquarium pump sends air through a dehumidifier (optional, see later). The air then bubbles through a water filled flask that is thermally insulated. Spray is trapped by a washing up scourer of twisted plastic ribbon. The emerging air is saturated but at a lower temperature than the room air, because the latent heat of evaporation is drawn from the air, as in a psychrometer. This air stream is then passed through a long (about 1 metre, depending on air flow) coil of copper pipe to bring it back to room temperature. During this process the relative humidity falls. The air then passes into the reaction vessel containing the object to be humidified.
The principle is very simple. We need to add methods for measuring and regulating RH and we should add some safety features.
Two thermometers are required.The RH is defined by the ratio of the saturation vapour pressures of water at the temperature of the water in the flask and at the temperature of the air in the humidification chamber. There is therefore no need for a direct RH measurement, except once, to check that saturation is actually achieved at the desired air flow (there will be more about RH measurement in a later article).
The air flow is first adjusted to the required value. The water temperature is then adjusted by changing the efficiency of the insulation. The apparatus illustrated can achieve any RH between 95% and 40%. It is, however, surprisingly difficult to achieve a high RH. If only a high RH is required and the room RH is stable, the drier can be omitted. A second saturator can be added, or a fan used to blow room air past the saturator.
The usual error is forgetting to top up the water, but we do not recommend automatic water level control. The central tube in the saturator functions as a pressure release valve. When the water level falls below the bottom of the tube the air will go up the tube instead of into the humidity chamber. The convolutions of the copper heat exchanger will ensure that the air will more easily escape through the straight tube in the saturator. To be quite sure, you can put a slight resistance to air flow at the entrance to the humidification chamber; a rubber flap with a very thin brass spring will do.
Guarding against exhaustion of the drier is more difficult. If silica gel is used it will fail gradually, so one must be really careless to cause a high RH this way. Molecular sieve is a more efficient drier but fails suddenly.

This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 License.